Share this
Authors
Yamamoto, Akira; Liu, Xueshi; Arashiba, Kazuya; Konomi, Asuka; Tanaka, Hiromasa; Yoshizawa, Kazunari; Nishibayashi, Yoshiaki; Yoshida, Hisao
Abstract
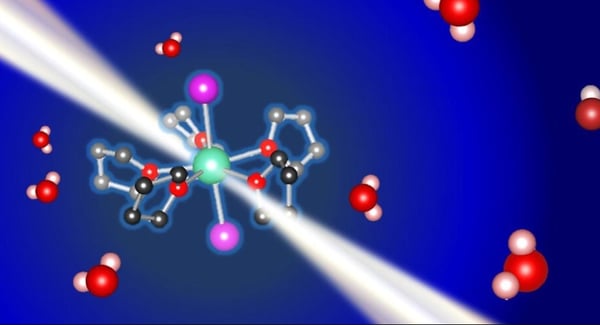
Chemoselective reductive conversion of organic and inorganic compounds has been developed by the combination of samarium(II) diiodide (SmI2) and water. Despite the extensive previous studies to elucidate the role of water in the reactivity of SmI2, the direct structural data of the reactive Sm2+–water complexes, SmI2(H2O)n, in an organic solvent–water mixture have not been reported experimentally so far. Herein, we performed the structure analysis of the Sm2+–water complex in tetrahydrofuran (THF) in the presence of water by in situ X-ray absorption spectroscopy using high-energy X-rays (Sm K-edge, 46.8 keV). The analysis revealed the dissociation of the Sm2+–I bonds in the presence of ≥ eight equivalents of water in the THF–water mixture. The origin of the peak shift in the UV/visible absorption spectra after the addition of water into SmI2/THF solution was proposed based on electron transitions simulated with time-dependent density-functional-theory calculations using optimized structures in THF or water. The obtained structural information provides the fundamental insights for elucidating the reactivity and chemoselectivity in the Sm2+–water complex system.
Inorganic Chemistry: https://pubs.acs.org/doi/full/10.1021/acs.inorgchem.2c03752