Share this
Authors
Kaneko Yudai, Yamatsugu Kenzo, Yamashita Takefumi, Takahashi Kazuki, Tanaka Toshiya, Aki Sho, Tatsumi Toshifumi, Kawamura Takeshi, Miura Mai, Ishii Masazumi, Ohkubo Kei, Osawa Tsuyoshi, Kodama Tatsuhiko, Ishikawa Shumpei, Tsukagoshi Masanobu, Chansler Michael, Sugiyama Akira, Kanai Motomu, Katoh Hiroto
Abstract
Antibody–mimetic drug conjugate is a novel noncovalent conjugate consisting of an antibody-mimetic recognizing a target molecule on the cancer cell surface and low-molecular-weight payloads that kill the cancer cells. In this study, the efficacy of a photo-activating antibody–mimetic drug conjugate targeting HER2-expressing tumors was evaluated in mice, by using the affibody that recognize HER2 (ZHER2:342) as a target molecule and an axially substituted silicon phthalocyanine (a novel potent photo-activating compound) as a payload. The first treatment with the photo-activating antibody–mimetic drug conjugates reduced the size of all HER2-expressing KPL-4 xenograft tumors macroscopically. However, during the observation period, relapsed tumors gradually appeared in approximately 50% of the animals. To evaluate the efficacy of repeated antibody–mimetic drug conjugate treatment, animals with relapsed tumors were treated again with the same regimen. After the second observation period, the mouse tissues were examined histopathologically. Unexpectedly, all relapsed tumors were eradicated, and all animals were diagnosed with pathological complete remission. After the second treatment, skin wounds healed rapidly, and no significant side effects were observed in other organs, except for occasional microscopic granulomatous tissues beneath the serosa of the liver in a few mice. Repeated treatments seemed to be well tolerated. These results indicate the promising efficacy of the repeated photo-activating antibody–mimetic drug conjugate treatment against HER2-expressing tumors.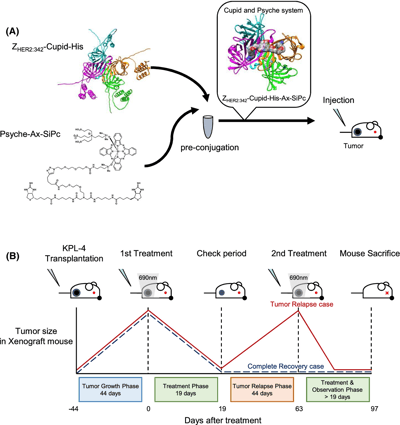
Cancer Science: https://onlinelibrary.wiley.com/doi/10.1111/cas.15565