Authors
Tao Yang, Yuki Mochida, Xueying Liu, Hang Zhou, Jinbing Xie, Yasutaka Anraku, Hiroaki Kinoh, Horacio Cabral & Kazunori Kataoka
Abstract
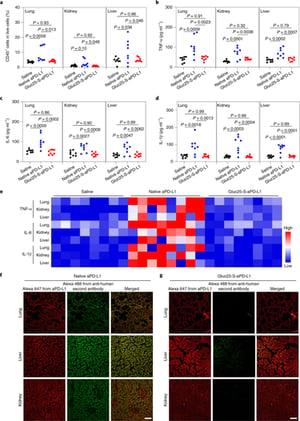
Because of the blood–tumour barrier and cross-reactivity with healthy tissues, immune checkpoint blockade therapy against glioblastoma has inadequate efficacy and is associated with a high risk of immune-related adverse events. Here we show that anti-programmed death-ligand 1 antibodies conjugated with multiple poly(ethylene glycol) (PEG) chains functionalized to target glucose transporter 1 (which is overexpressed in brain capillaries) and detaching in the reductive tumour microenvironment augment the potency and safety of checkpoint blockade therapy against glioblastoma. In mice bearing orthotopic glioblastoma tumours, a single dose of glucosylated and multi-PEGylated antibodies reinvigorated antitumour immune responses, induced immunological memory that protected the animals against rechallenge with tumour cells, and suppressed autoimmune responses in the animals’ healthy tissues. Drug-delivery formulations leveraging multivalent ligand interactions and the properties of the tumour microenvironment to facilitate the crossing of blood–tumour barriers and increase drug specificity may enhance the efficacy and safety of other antibody-based therapies.
Nature Biomedical Engineering:https://www.nature.com/articles/s41551-021-00803-z