Authors
Shohei Tada, Hiroka Kinoshita, Nagomu Ochiai, Anand Chokkalingam, Peidong Hu, Noriko Yamauchi, Yoshio Kobayashi, Kenta Iyoki
Abstract
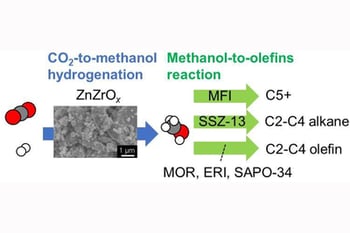
The development of methodologies for CO2 utilization is in high demand worldwide. Here, we propose bifunctional tandem catalysts of ZnZrOx (for CO2-to-methanol hydrogenation) and a series of solid acid catalysts (for subsequent methanol conversion to light olefins). As solid acid catalysts, we used zeolites and silicoaluminophosphates with different topologies, MOR, FER, MFI, ∗BEA, CHA, and ERI, confirmed by X-ray diffraction, electron microscopy, and nitrogen adsorption-desorption. They also showed the corresponding acid properties examined by ammonia adsorption. The tandem catalysts realized a one-pass synthesis of lower olefins, while no hydrocarbons were obtained using ZnZrOx only. According to the reaction test and ammonia adsorption, there seems to be no correlation between product yields and acid strength. The pore sizes and channel dimensionality of zeolites influence the selectivity of products; zeolites with small pores, such as MOR, SAPO-34 and ERI, are promising, while zeolites with larger pores, such as MFI, produce heavier hydrocarbons. The results provide new insight into the design of innovative catalysts for CO2 utilization.
International Journal of Hydrogen Energy : https://www.sciencedirect.com/science/article/pii/S0360319921034820?via%3Dihub