Share this
Authors
Hiroki Takezawa, Kosuke Shitozawa, Makoto Fujita
Abstract
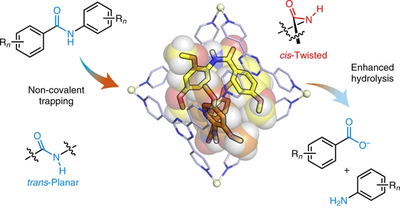
When an amide group is distorted from its planar conformation, the conjugation between the nitrogen lone pair and the π* orbital of the carbonyl is disrupted and the reactivity towards nucleophiles is enhanced. Although there are several reports on the synthesis of activated twisted amides, amide activation through mechanical twisting is much less common. Here, we report twisted amides that are stabilized through their inclusion in a self-assembled coordination cage. When secondary aromatic amides are included in a Td-symmetric cage, the cis-twisted conformation is favoured over the trans-planar one—as evidenced by single-crystal X-ray diffraction analysis—revealing that the amide can twist by up to 34°. As a consequence of this distortion, the hydrolysis of amides is significantly accelerated upon inclusion.
Nature Chemistry:https://www.nature.com/articles/s41557-020-0455-y
These Related Stories