Share this
Authors
Keiju Wachi, Tomohiro Yabe, Takaaki Suzuki, Kentaro Yonesato, Kosuke Suzuki, Kazuya Yamaguchi
Abstract
Direct oxidative conversion of methane (CH4) into useful C1 productsremains challenging due to the low reactivity of CH4 and its facile overoxidation into carbon dioxide (CO2) under high temperature conditions. Iron oxide (FeOx) cluster catalysts are promising because of their low activation energy for CH4 oxidation. However, preliminary results revealed that FeOx clusters were easily aggregated and deactivated under CH4 oxidation conditions at 873 K. In this study, we used a diiron-introduced polyoxometalate as a precursor to form thermally stable FeOx subnanoclusters on SiO2, which selectively converted CH4 into formaldehyde (HCHO) and carbon monoxide (CO) (CH4 conversion, 2.3%; HCHO and CO selectivity, 87% at 873 K after 1 h). The FeOx subnanocluster catalyst maintained catalytic activity even after 72 h. Various characterizations, such as STEM, X-ray absorption spectroscopy, and X-ray diffraction, revealed that the in situ formed FeOx subnanoclusters were stabilized by WOx nanoclusters originating from the polyoxometalate frameworks.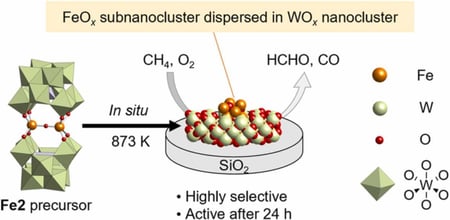
Applied Catalysis B: Environmental: https://doi.org/10.1016/j.apcatb.2022.121420