Share this
Authors
Jing Wang, Yuka Yashiro, Yuriko Sakaguchi, Tsutomu Suzuki, and Kozo Tomita
Abstract
Contact-dependent growth inhibition is a mechanism of interbacterial competition mediated by delivery of the C-terminal toxin domain of CdiA protein (CdiA–CT) into neighboring bacteria. The CdiA–CT of enterohemorrhagic Escherichia coli EC869 (CdiA–CTEC869) cleaves the 3′-acceptor regions of specific tRNAs in a reaction that requires the translation factors Tu/Ts and GTP. Here, we show that CdiA–CTEC869 has an intrinsic ability to recognize a specific sequence in substrate tRNAs, and Tu:Ts complex promotes tRNA cleavage by CdiA–CTEC869. Uncharged and aminoacylated tRNAs (aa-tRNAs) were cleaved by CdiA–CTEC869 to the same extent in the presence of Tu/Ts, and the CdiA–CTEC869:Tu:Ts:tRNA(aa-tRNA) complex formed in the presence of GTP. CdiA–CTEC869 interacts with domain II of Tu, thereby preventing the 3′-moiety of tRNA to bind to Tu as in canonical Tu:GTP:aa-tRNA complexes. Superimposition of the Tu:GTP:aa-tRNA structure onto the CdiA–CTEC869:Tu structure suggests that the 3′-portion of tRNA relocates into the CdiA–CTEC869 active site, located on the opposite side to the CdiA–CTEC869 :Tu interface, for tRNA cleavage. Thus, CdiA–CTEC869 is recruited to Tu:GTP:Ts, and CdiA–CT:Tu:GTP:Ts recognizes substrate tRNAs and cleaves them. Tu:GTP:Ts serves as a reaction scaffold that increases the affinity of CdiA–CTEC869 for substrate tRNAs and induces a structural change of tRNAs for efficient cleavage by CdiA–CTEC869.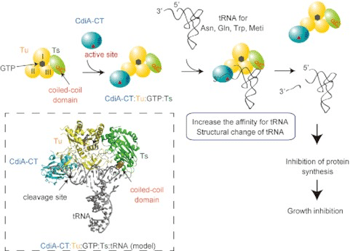
Nucleic Acids Research: https://academic.oup.com/nar/article/50/8/4713/6565320