Share this
Authors
Toshihiro Okamoto, Masato Mitani, Craig P. Yu, Chikahiko Mitsui, Masakazu Yamagishi, Hiroyuki Ishii, Go Watanabe, Shohei Kumagai, Daisuke Hashizume, Shota Tanaka, Masafumi Yano, Tomokatsu Kushida, Hiroyasu Sato, Kunihisa
Sugimoto, Takashi Kato, and Jun Takeya
Abstract
Toward the development of high-performance organic semiconductors (OSCs), carrier mobility is the most important requirement for next-generation OSC-based electronics. The strategy is that OSCs consisting of a highly extended π-electron core exhibit two-dimensional (2D) aggregated structures to offer effective charge transport. However, such OSCs, in general, show poor solubility in common organic solvents, resulting in limited solution processability. This is a critical trade-off between the development of OSCs with simultaneous high carrier mobility and suitable solubility. To address this issue, herein, five-membered ring-fused selenium-bridged V-shaped binaphthalene with decyl substituents (C10–DNS–VW) is developed and synthesized by an efficient method. C10–DNS–VW exhibits significantly high solubility for solution processes. Notably, C10–DNS–VW forms a one-dimensional π-stacked packing motif (1D motif) and a 2D herringbone (HB) packing motif (2D motif), depending on the crystal growth condition. On the other hand, the fabrication of thin films by means of both solution process and vacuum deposition techniques forms only the 2D HB motif. External stress tests such as heating and exposure to solvent vapor indicated that 1D and 2D motifs could be synergistically induced by the total balance of intermolecular interactions. Finally, the single-crystalline films of C10–DNS–VW by solution process exhibit carrier mobility up to 11 cm2 V–1 s–1 with suitable transistor stability under ambient conditions for more than two months, indicating that C10–DNS–VW is one of the most promising candidates for breaking the trade-off in the field of solution-processed technologies.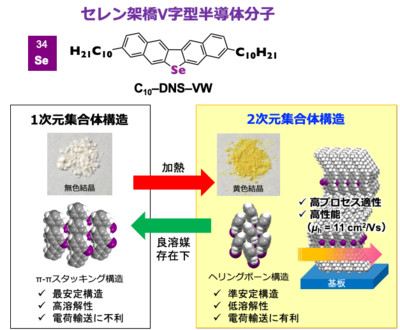
Journal of the American Chemical Society:https://pubs.acs.org/doi/10.1021/jacs.0c05522
These Related Stories