Share this
Authors
Ryosuke Ueki, Satoshi Uchida, Naoto Kanda, Naoki Yamada, Ayaka Ueki, Momoko Akiyama, Kazuko Toh, Horacio Cabral, and Shinsuke Sando
Abstract
Although growth factors have great therapeutic potential because of their regenerative functions, they often have intrinsic drawbacks, such as low thermal stability and high production cost. Oligonucleotides have recently emerged as promising chemical entities for designing synthetic alternatives to growth factors. However, their applications in vivo have been recognized as a challenge because of their susceptibility to nucleases and limited distribution to a target tissue. Here, we present the first example of oligonucleotide-based growth factor mimetics that exerts therapeutic effects at a target tissue after systemic injection. The aptamer was designed to dimerize a growth factor receptor for its activation and mitigated the progression of Fas-induced fulminant hepatitis in a mouse model. This unprecedented functionality of the aptamer can be reasonably explained by its high nuclease stability and migration to the liver parenchyma. These mechanistic analyses provided insights for the successful application of aptamer-based receptor agonists.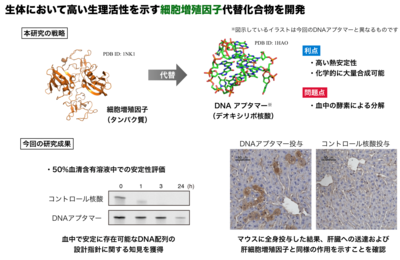
Science Advances:https://advances.sciencemag.org/content/6/14/eaay2801.full
These Related Stories