Share this
Authors
Nobutoshi Ota, Yusuke Yonamine, Takuya Asai, Yaxiaer Yalikun, Takuro Ito, Yasuyuki, Ozeki, Yu Hoshino and Yo Tanaka
Abstract
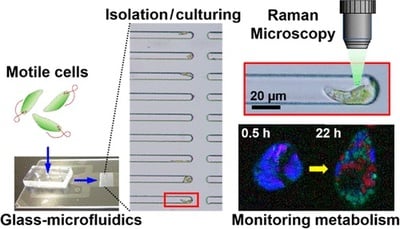
Time-course analysis of single cells is important to characterize heterogeneous activities of individual cells such as the metabolic response to their environment. Single-cell isolation is an essential step prior to time-course analysis of individual cells by collecting, culturing, and identifying multiple single-cell targets. Although single-cell isolation has been performed by various methods previously, a glass microfluidic device with semiclosed microchannels dramatically improved this process with its simple operation and easy transfer for time-course analysis of identified single cells. This study demonstrates isolating single cells of the highly motile microalgae, Euglena gracilis, by semiclosed microchannels with liquid flow only. The isolated single cells were identified in isolating channels and continuously cultured to track, by Raman microscopy, for the formation of subcellular granules composed of polysaccharide paramylon, a unique metabolite of E. gracilis, generated through photosynthesis. Through low-temperature glass bonding, a thin glass interface was incorporated to the microfluidic device. Thus, the device could perform the direct measurements of cultured single cells at high magnification by Raman microscopy with low background noise. In this study, the first demonstration of sequential monitoring of paramylon biogenesis in a single identified E. gracilis cell is shown.
Analytical Chemistry:https://pubs.acs.org/doi/10.1021/acs.analchem.9b01007