Authors
Hiroaki Kinoh, Yutaka Miura, Tsukasa Chida, Xueying Liu, Kazue Mizuno, Shigeto Fukushima, Yosuke Morodomi, Nobuhiro Nishiyama, Horacio Cabral, and Kazunori Kataoka
Abstract
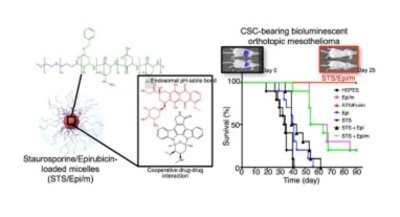
Nanomedicines capable of control over drug functions have potential for developing resilient therapies, even against tumors harboring recalcitrant cancer stem cells (CSCs). By coordinating drug interactions within the confined inner compartment of core-shell nanomedicines, we conceived multicomponent nanomedicines directed to achieve synchronized and synergistic drug cooperation within tumor cells as a strategy for enhancing efficacy,overcoming drug resistance and eradicating CSCs. The approach was validated by using polymeric micellar nanomedicines co-incorporating the pan-kinase inhibitor staurosporine (STS), which was identified as the most potent CSC-inhibitor from a panel of signaling-pathway inhibitors, and the cytotoxic agent epirubicin (Epi), through rationally contriving the affinity between the drugs. The micelles released both drugs simultaneously, triggered by acidic endosomal pH, attaining concurrent intracellular delivery, with STS working as a companion for Epi, down-regulating efflux transporters and resistance mechanisms induced by Epi. These features prompted the nanomedicines to eradicate orthotopic xenografts of Epi-resistant mesothelioma bearing CSC sub-population.
Recalcitrant cancer stem cells (CSCs) have been identified in many cancer types, and have been associated with metastasis, resistance to therapy, and the eventual relapse of the disease. With traditional chemotherapeutics being futile against the CSC population, the effective combination of CSC inhibitors with cytotoxic therapies emerges as crucial for achieving robust responses capable of long-term disease-free survival. Accordingly, CSCs have shown deregulation in several pathways involved in the control of self-renewal and differentiation, such as PI3K/Akt, Wnt/β-catenin, PTEN, NF-κB, JAK/STAT, hedgehog, Notch, Bcl-2, which represent promising targets for the development of clinically useful CSC inhibitors. Besides, the crosstalk between these CSCs-related signaling pathways and their influence on tumor progression offer extra opportunity for implementation of multi-targeted therapies, in particular for synergistic combination with cytotoxic treatments. However, as many of these pathways are found in normal stem cells, the complete eradication of CSCs with such inhibitors compels a challenging task due to the underlying risk of damage to healthy tissues, and the toxicity of CSC inhibitors combined with other anticancer agents could be rendered intolerable. Therefore, effective strategies should be designed for sufficient selectivity of these inhibitors toward CSCs.
Nanomedicine has demonstrated great potential for developing safe and targetedstrategies against solid tumors. Thus, nanomedicine approaches involving the targeting of both CSCs inhibitors and cytotoxic drugs to tumor tissues could serve as an effective way for controlling side effects, while improving treatment outcomes, eventually against recalcitrant cancers. However, beyond the mere co-incorporation of drugs, such nanomedicines should precisely tailor the intracellular interplay of CSCs inhibitors and cytotoxic agents to actually achieve synchronized activities and potentiate cooperative synergistic efficacies. For this purpose, drug-loaded polymeric micelles, i.e. core-shell nano-assemblies, appear as a translationable nanomedicine modality with the capacity for effectively controlling the interaction of therapeutics through their sheltered core structure.
Herein, by exploiting the molecular interaction between drug molecules, we co-loaded CSCs inhibitors and cytotoxic drugs into polymeric micelles, and profited from the compartmentalized architecture of the micelles for maintaining this interaction until reaching the intracellular space of cancer cells and CSCs in tumors, where the release of both drugs is triggered by endosomal pH to attain coordinated therapeutic effects. Staurosporine (STS), a broadly selective and potent protein kinase inhibitor, was selected as the CSCs inhibitor in this study, after being identified as the most potent agent against the CSC fraction from a panel of inhibitors of signaling pathways. The affinity of STS, as well as the synergistic efficacy, with the anthracycline epirubicin (Epi) was then exploited to encompass STS into the core of Epi-loaded polymeric micelles (Epi/m), which are being studied in Phase I clinical studies, for intracellular synchronization of therapeutic effects. Our results indicated that these multicomponent STS/Epiloaded micelles (STS/Epi/m) eliminated orthotopic xenografts of Epi-resistant mesothelioma, a lethal cancer of the mesothelium, through facilitated co-delivery to tumor tissues, triggered and coordinated release of both drugs at endosomal pH, cooperative drug interactions, eradication of CSC sub-population and reversal of drug resistance through ABC transporter inhibition.
Abstract URL:http://pubs.acs.org/doi/pdf/10.1021/acsnano.6b00900
You May Also Like