PRESS RELEASE
- Research
- 2016
Approaching the Limit of Zeolite Crystallization: Continuous Flow Synthesis of ZSM-5 on the Order of Seconds
Authors
Zhendong Liu, Kotatsu Okabe,Chokkalingam Anand,Yasuo Yonezawa, Jie Zhu, Hiroki Yamada, Akira Endo, Yutaka Yanaba,
Takeshi Yoshikawa, Koji Ohara, Tatsuya Okubo, and Toru Wakihara
Abstract
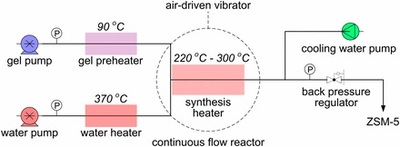
The hydrothermal synthesis of zeolites carried out in batch reactors takes a time so long (typically, on the order of days) that the crystallization of zeolites has long been believed to be very slow in nature. We herein present a synthetic process for ZSM-5, an industrially important zeolite, on the order of seconds in a continuous flow reactor using pressurized hot water as a heating medium. Direct mixing of a well-tuned precursor (90 °C) with the pressurized water preheated to extremely high temperature (370 °C) in the millimeter-sized continuous flow reactor resulted in immediate heating to high temperatures (240–300 °C); consequently, the crystallization of ZSM-5 in a seed-free system proceeded to completion within tens of or even several seconds. These results indicate that the crystallization of zeolites can complete in a period on the order of seconds. The subtle design combining a continuous flow reactor with pressurized hot water can greatly facilitate the mass production of zeolites in the future.
PNAS URL: http://www.pnas.org/content/early/2016/11/23/1615872113

